Biological mechanisms underlying remineralization of vascular plant material in the presence of diatoms
Abstract
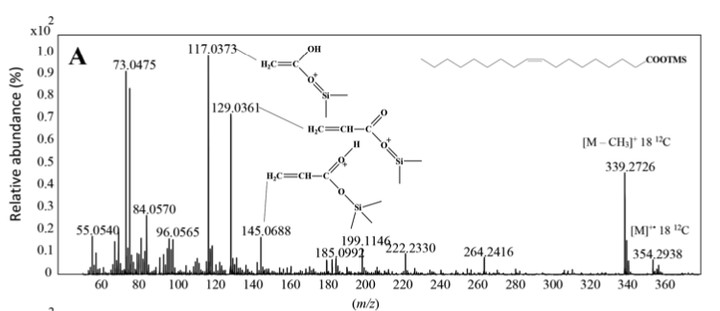
Priming effects that stimulate increased degradation of refractory organic matter by microorganisms following fresh organic matter input is a well-known phenomenon in terrestrial environments but remains controversial in marine environments. We used a combination of chem- ical (gas chromatography-EI quadrupole time of flight mass spectrometry) and molecular biology (DNA stable-isotope probing [DNA-SIP]) methods to trace the fate of terrestrially derived partic- ulate organic matter (TPOM) and the response of the marine microbial community to fresh organic matter inputs. We tested the potential for priming effects among a mixture of marine and terrestrial microbial assemblages, amended either with only 13C-labelled TPOM (13C-Avena sativa) or with an addition of 12C-Skeletonema costatum (a marine diatom) as a labile co-substrate within the range of diatom concentrations found in estuaries. We monitored 13C-labelled TPOM lipid tracers (long-chain fatty acids, n-alkan-1-ols, phytol, sitosterol, β-amyrin and components of cutins) throughout a 42 d incubation experiment. Comparisons with controls carried out without diatom addition showed faster decay of phytol, n-alkan-1-ols, and components of cuticular waxes in the presence of diatoms, while fatty acids and sitosterol were unaffected. Bacteria belonging to the Bacteroidota phylum (mainly Flavobacteria and Cytophaga) were the dominant microbes involved in priming-induced TPOM degradation in the incubation treatments. Sphingomonadales and Rhizobiales, capable of lignin and hemicellulose degradation, also contributed to the degra- dation of TPOM but did not seem to contribute to priming effects related to increased diatom abundance. These lab-based results demonstrate direct evidence that priming of TPOM occurred selectively via a consortium of microbes.